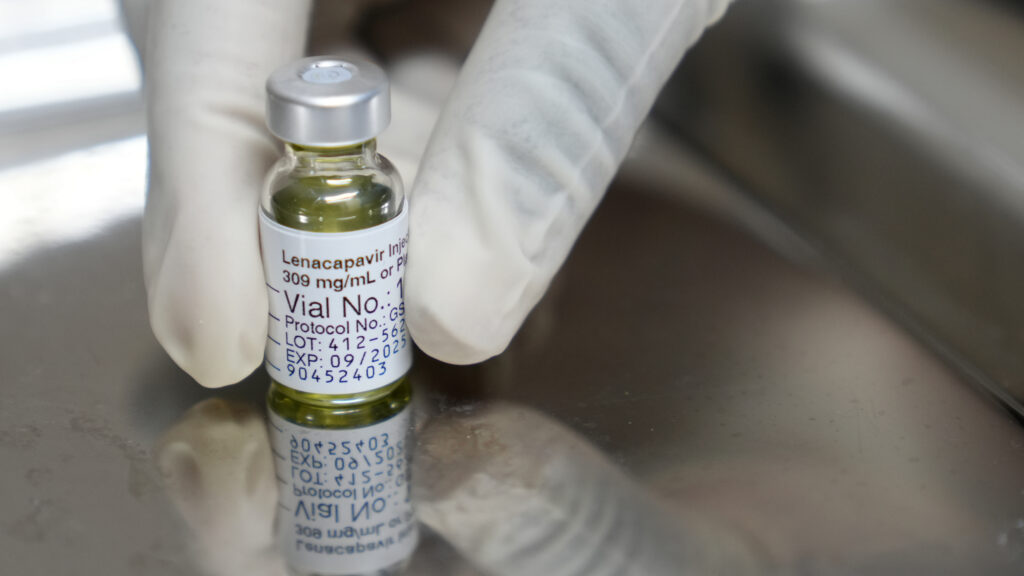
The U.S. Department of State has begun the distribution of a groundbreaking HIV prevention drug to select countries, with the recent delivery of approximately 1,000 doses of the Gilead Sciences medication to Zambia and Eswatini. This marks the initial phase of a distribution plan announced in fall 2022, aimed at combating the high rates of HIV infection in vulnerable regions.
Gilead Sciences, the manufacturer of the drug, is providing it at cost as part of a broader initiative to enhance global health responses. The company is actively pursuing regulatory approval for its medication in over a dozen other sub-Saharan nations, which continue to experience alarming rates of HIV infection. This region remains the epicenter of the ongoing AIDS pandemic, highlighting the urgent need for effective prevention measures.
During a media briefing, Jeremy Lewin, a senior official at the U.S. Department of State, emphasized the significance of this initiative. He stated, “I think this is a prime example of what we can do to accelerate progress on global health through championing American innovation and American enterprise.” This perspective underscores the U.S. government’s commitment to leveraging its resources and partnerships to address global health challenges.
While the initiative has garnered attention for its potential impact, it notably excludes South Africa, which has one of the highest HIV prevalence rates in the world. Critics have raised concerns regarding the omission, calling for a more inclusive approach that encompasses all countries grappling with the HIV epidemic. The absence of South Africa from the initial distribution list raises questions about equitable access to vital health resources.
As the U.S. government continues to highlight its efforts in global health, the effectiveness of this program will depend on the broader engagement with affected nations. The response from countries like Zambia and Eswatini will be closely monitored as they incorporate the medication into their public health strategies. Meanwhile, Gilead Sciences remains dedicated to expanding access to its HIV prevention drug across the continent, with a focus on regulatory approvals that can facilitate wider distribution.
The challenges posed by the HIV epidemic require a concerted global effort, and initiatives like this one reflect a step towards addressing the pressing health needs of vulnerable populations. As the situation evolves, the implications of this distribution plan will be critical in informing future health interventions and policies aimed at curbing the spread of HIV in sub-Saharan Africa.