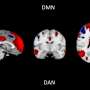
Researchers from Université Savoie Mont Blanc, Radboud University, and the University of Oxford have identified a new fMRI biomarker that may provide insights into cognitive decline associated with Alzheimer’s disease. This study reveals a significant attenuation in the brain’s intrinsic anticorrelation between the default mode network (DMN) and dorsal attention network (DAN), suggesting a reliable indicator of Alzheimer’s-related cognitive decline that operates independently of tau pathology and educational cognitive reserve.
Despite ongoing research, Alzheimer’s disease still lacks a definitive biomarker capable of pinpointing the onset of abnormal protein accumulation and subsequent network failures. Current understanding of the disease’s progression remains fragmented, with mechanisms linking amyloid and tau pathology, vascular lesions, and neurodegeneration. Notably, beta-amyloid accumulation can occur years before clinical symptoms manifest, indicating a spectrum of progression rather than a distinct onset of dementia.
Understanding Network Interactions
The human brain comprises various neurocognitive networks that can operate in cooperation or competition. Resting-state functional MRI (fMRI) has advanced our understanding of how these networks interact without the need for specific tasks. The DMN and DAN typically exhibit an inverse relationship: when one is active, the other is deactivated. This dynamic is evident during daydreaming or self-reflection, where the DMN is engaged, compared to focused tasks that activate the DAN.
Prior studies have indicated that alterations in resting-state functional connectivity within these networks are linked to cognitive decline in healthy adults. Research has proposed that a “functional imbalance” could underlie neurodegenerative pathology, with the interaction between DMN and DAN possibly serving as a systems-level signature related to cognitive decline.
The latest study, titled “The intrinsic connectivity between the default mode and dorsal attention networks is an independent fMRI biomarker of Alzheimer’s disease pathology burden,” published in NeuroImage, involved a comprehensive analysis of MRI, PET amyloid and tau imaging, alongside cognitive assessments from 182 participants in the Alzheimer’s Disease Neuroimaging Initiative (ADNI) 3 cohort.
Key Findings on Cognitive Performance
Researchers focused on how DMN-DAN anticorrelation correlates with Alzheimer’s pathology burden, specifically amyloid and tau levels. They measured resting brain activity across large-scale networks, assessing how these networks interacted over time. A notable finding was that participants with high amyloid burdens and cognitive impairments exhibited reduced separation between the DMN and DAN signals, indicating less distinct network activity.
This diminished separation can hinder cognitive functions, making tasks more susceptible to interference, thus complicating attention, memory retrieval, and task coordination. The study found that weaker anticorrelation between these networks corresponded with lower cognitive scores, where approximately 5% of the variance in cognitive performance could be attributed to this single fMRI measure.
Furthermore, analyses linked the DMN-DAN connectivity to scores on the Mini-Mental State Examination and various cognitive tests, reinforcing the association across multiple cognitive domains. Interestingly, the level of education, often used as a proxy for cognitive reserve, did not alter this relationship.
The implications of these findings suggest a departure from traditional views of Alzheimer’s pathology. The authors propose a multifaceted model of cognitive decline involving various factors, including vascular disease and stress, rather than a linear progression driven solely by tau-related neurodegeneration.
Future research directions highlighted in the study advocate for longitudinal and interventional studies to determine whether network dysfunction precedes or follows abnormal protein accumulation. Additionally, developing personalized risk models that integrate DMN-DAN connectivity patterns, pathology measures, and lifestyle factors could enhance understanding and treatment strategies for Alzheimer’s disease.
This research sheds light on the intricate nature of cognitive decline in Alzheimer’s, positioning resting-state fMRI as a potential functional marker that captures mechanisms of cognitive dysfunction that are not entirely explained by tau pathology or cognitive reserve.